July 10, 2024 (press release) –
- Company Committed to Support Global Health and Customers
- BD Taking all Available Measures to Mitigate Supplier Issue
, July 10, 2024 /PRNewswire/ -- BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today issued the following statement related to a U.S. FDA letter to health care providers regarding the supply of blood culture vials:
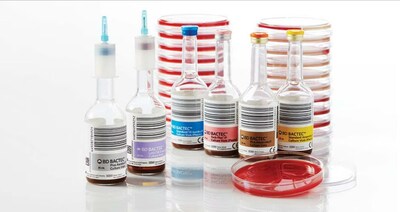
Nikos Pavlidis, worldwide president of BD Diagnostic Solutions said, "BD is experiencing reduced availability of blood culture vials from our supplier. We understand the critical role that blood culture testing plays in diagnosing and treating infections and are taking all available measures to address this important issue, including providing the supplier our manufacturing expertise, using air shipments, modifying BD manufacturing schedules for rapid production, and collaborating with the U.S. Food and Drug Administration to review all potential options to mitigate delays in supply. As an additional stopgap measure, our former supplier of glass vials will restart production to help fill the intermittent gap in supply.
"We take our responsibility to support health care providers and patients very seriously and, while we are able to meet the majority of global demand, we are taking all available measures to address this important issue. BD BACTEC™ blood culture vials are being allocated manually so we can best mitigate any impact to patient care. Customers have been advised to prioritize the use of blood culture vials based on clinical needs and following guidelines, such as the most recent update from the Infectious Disease and Society of America and/or the World Health Organization.
"While this supplier issue is not expected to have a material financial impact on BD, we are fully focused on returning the supply of blood culture vials to normal levels."
More information on BD BACTEC™ blood culture vials supply can be found at BDBACTEC-update.com. This matter is not related to BD Vacutainer® blood collection tubes.
About BD
BD is one of the largest global medical technology companies in the world and is advancing the world of health by improving medical discovery, diagnostics and the delivery of care. The company supports the heroes on the frontlines of health care by developing innovative technology, services and solutions that help advance both clinical therapy for patients and clinical process for health care providers. BD and its 70,000 employees have a passion and commitment to help enhance the safety and efficiency of clinicians' care delivery process, enable laboratory scientists to accurately detect disease and advance researchers' capabilities to develop the next generation of diagnostics and therapeutics. BD has a presence in virtually every country and partners with organizations around the world to address some of the most challenging global health issues. By working in close collaboration with customers, BD can help enhance outcomes, lower costs, increase efficiencies, improve safety and expand access to health care. For more information on BD, please visit bd.com or connect with us on LinkedIn at www.linkedin.com/company/bd1/ and Twitter @BDandCo.
* All content is copyrighted by Industry Intelligence, or the original respective author or source. You may not recirculate, redistrubte or publish the analysis and presentation included in the service without Industry Intelligence's prior written consent. Please review our terms of use.
